Cambridge, MA, March 17, 2026 – Lysoway Therapeutics is pleased to announce that Dr. Valerie Cullen, Senior Vice President of Research and Translation, will present both an oral presentation and a poster at the 2026 ADPD Meeting in Copenhagen, Denmark, a premier global forum for scientific and clinical advances in Alzheimer’s and Parkinson’s diseases.
Dr. Cullen’s oral presentation, “The TRPML1 Agonist, LW-1017, Dose-Dependently Ameliorates Disease Pathology and Improves Behavior in Mouse Models of Alzheimer’s Disease and Parkinson’s Disease,” will be delivered on Saturday, March 21, 2026, in Symposium 6060 (Emerging Therapies Across AD, PD, and LBD) at 3:55 p.m.
She will also present a poster, “Development of a Potent, Brain-Penetrant TMEM175 Agonist for the Treatment of Parkinson’s Disease,” on Wednesday, March 18, and Thursday, March 19, 2026.
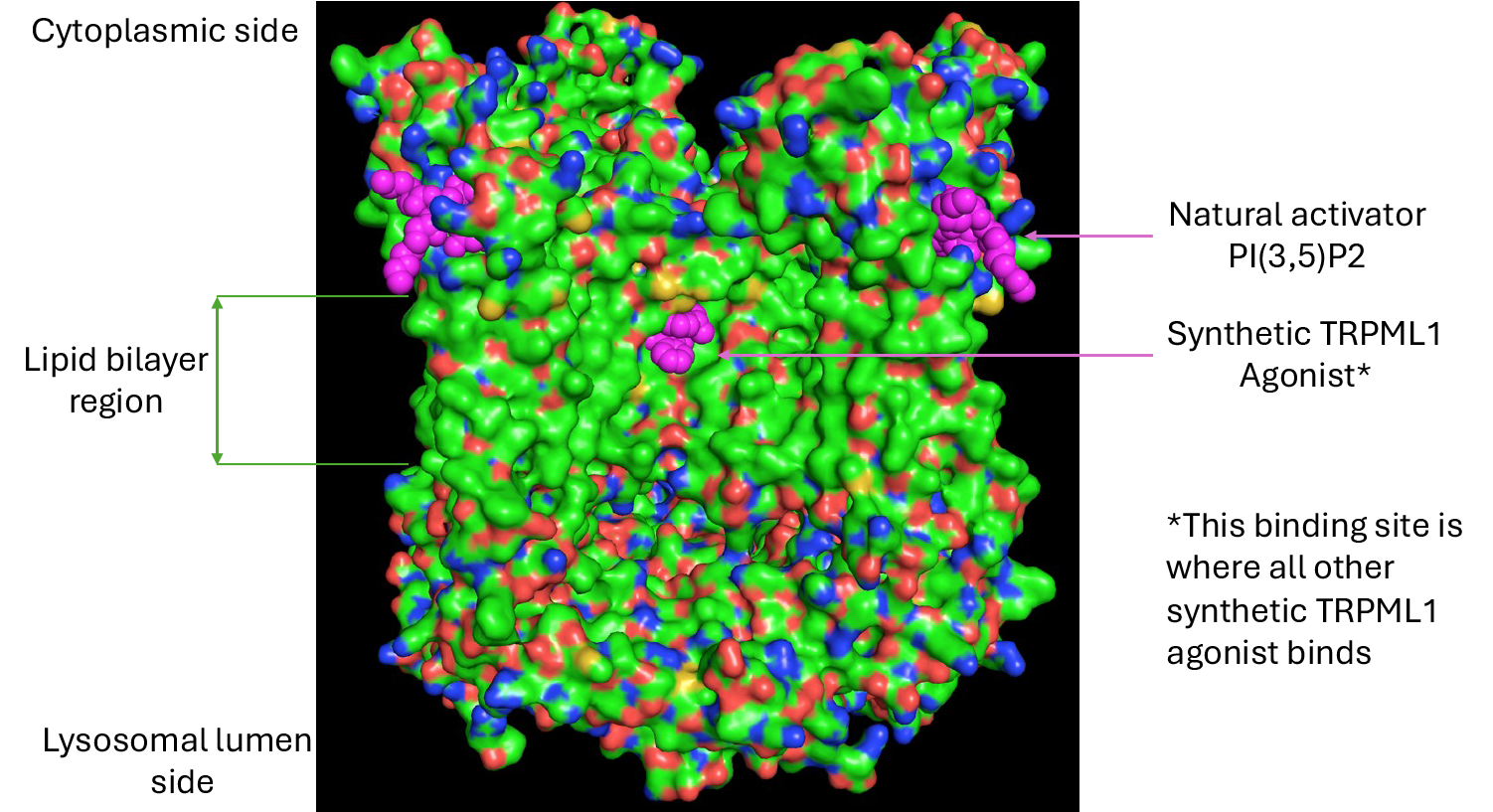
These presentations will feature Lysoway’s preclinical advances in TRPML1 and TMEM175 agonists, including data supporting lysosomal ion channels as promising therapeutic targets in neurodegenerative disease.
“Lysoway is honored to share our latest scientific progress at the 2026 ADPD Meeting,” said Dr. Yongchang Qiu, Founder and CEO of Lysoway Therapeutics, Inc. “These presentations reflect the strength of our lysosomal ion channel platform and our commitment to advancing differentiated therapies for neurodegenerative diseases.”
For more information about the conference, visit the 2026 ADPD Meeting website. To learn more about Lysoway’s platform and programs, please visit www.lysoway.com.
About Lysoway Therapeutics
Lysoway Therapeutics is a biotechnology company pioneering therapeutics targeting lysosomal ion channels to counteract age-related neurodegeneration. Leveraging structure-based drug design, high-resolution cryo-EM, and proprietary insights into lysosomal biology, Lysoway is developing highly brain-penetrant small-molecule agonists of TRPML1 and TMEM175 to restore coordinated autophagy–lysosomal function and cellular homeostasis. For more information, visit www.lysoway.com.
Contact Info
info@lysoway.com
www.lysoway.com
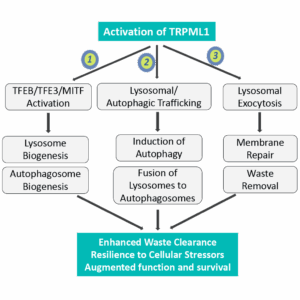 Figure 1.
Figure 1.